

Introducing Our State-of-the-Art Human Factors Study Facility
30 July 2024
We are pleased to announce an enhancement to our human factors capabilities with the introduction of our new dedicated human…

Pharmaceutical Industry Sustainability Targets, One Year On
22 May 2024
In the October 2022 “Drug Delivery & Environmental Sustainability” issue of ONdrugDelivery, We discussed climate-related targets set by 10 major pharmaceutical companies, along…
Shaping the future of healthcare: Data, AI and Us
8 May 2024
Mike Beck, our Head of Electronics spent some time in Boston at the Device Talks Conference last week. Here are…
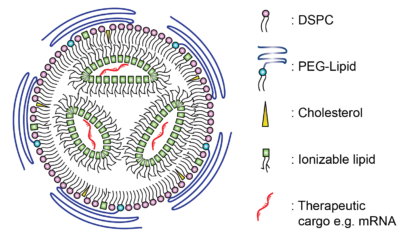
Bubble-Wrapped Remedies: How Lipid-Encased Therapeutics Could Transform Inhalation
30 April 2024
Introduction In a post-COVID-19 era we have witnessed a notable shift in vaccine development with the advent of lipid nanoparticle…

Springboard Pro to Host Networking Event for Startups in the Medical Device and Diagnostic Sphere
24 April 2024
Springboard Pro, a leading engineering consultancy, is pleased to announce its upcoming networking event tailored specifically for startups operating in…

Lab-On-Chip Devices: Materials Considerations
18 March 2024
Material selection and testing can often be one of the critical challenges to developing microfluidic devices for the healthcare and…

Bridging the gap: Adapting hospital equipment for home use
5 February 2024
The healthcare landscape is evolving, and one of the significant shifts is the increasing emphasis on home-based care. As technology…

The final frontier to improving low-pain injections
25 January 2024
Most people dislike injections. In some cases, that aversion comes from the pain experienced upon injection, and this discomfort can…

From Concept to Cure: Mastering Liquid Silicone Rubber for Medical Device Design
23 January 2024
Liquid silicone rubber (LSR) is frequently used in implants due to its bio-inert nature, meaning that it has a very…






