Connected drug delivery
16 July 2018
The need for connected drug delivery devices
Most people would agree that drug delivery devices have improved in recent years, particularly with the increased focus on usability (or ‘human factors’). However, adherence remains a challenge and payers are looking for ways to increase the cost-effectiveness of healthcare. One strategy for managing both issues is connecting drug delivery devices to the internet.
The potential benefits of connected drug delivery devices have been much discussed and can be summarised for stakeholders as follows:
- Patient
- Reminders
- Training
- Evidence for incentives
- Hawthorne effect
- Peer support
- Carer
- Reminders
- Training
- Payer
- (Non)adherence data
- Reduced costs (50% of patients suffering chronic illness do not take their medication as prescribed, costing US $100 billion to $300 billion annually in avoidable direct healthcare costs in the US alone [1])
- Healthcare professional
- (Non)adherence data
- Additional support for least adherent
- Adverse events
- Healthcare provider, or regulatory authority
- (Non)adherence data
- Adverse events
- Clinical trial data (pre and post market)
- Population trends
- Pharma
- Adverse events
- (Non)adherence data
- Clinical trial data (pre and post market)
- Reimbursement evidence
- Market understanding
- Product and training improvements
- Increased sales by increased adherence. (Non-adherence causes US $637 billion lost pharma revenue annually [2])
The disadvantages of connecting drug delivery devices to the internet are:
- Increased cost of devices and infrastructure.
- Potential increase in usability risks.
- Reliability risks due to technical complexity.
- Concerns over data privacy and robustness against hacking and malware.
- Unclear regulatory landscape due to the unfamiliarity of the technologies in the regulatory context.
- Environmental concerns for disposal of electronic waste.
Nevertheless, for some drugs and indications the advantages are compelling, so we should look at how connectivity might be implemented in a drug delivery device.
Device strategies
There are 3 main strategies for implementing connectivity in a drug delivery device:
- An add-on, typically to an existing design.
- An upgrade, which is integrated but does not change the core functionality or use case.
- Built-in, which can change the core functionality and use case.
Many pharmaceutical companies and drug delivery device manufacturers already have devices either on the market or in late stage development.
An industry survey of inhalers, for example, shows various companies using one or more of the three strategies. Note that we see the same trends in the injector industry.
Add-ons
Add-ons have the advantages that:
- They can be added to existing devices, in most cases without interfering with the existing device function.
- They could be reusable even if the existing device is disposable.
- Patients could pay for the add-on themselves, whereas the existing drug delivery device might be paid for by an insurer or national health service.
- There is less development risk due to limited revalidation of the existing device.
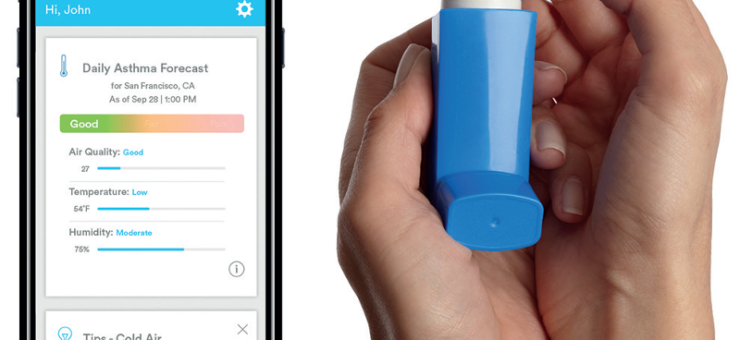
Propeller and Adherium are notable because they support a wide range of existing inhalers.

Propeller Health’s smart phone app, and inhaler add-on

Adherium’s add-ons fitted to various commercial inhalers, and Adherium’s smart phone app
Cohero provides a spirometer so patients can measure their lung function. This combination of drug delivery device and diagnostic device is powerful because it can provide direct disease management results to help patients and clinicians assess progress and can enable payment-by-results rather than pay-per-dose. Linking delivery devices with diagnostics was pioneered by the insulin pens (and to a greater degree, insulin pumps) and blood glucose meters used for diabetes. The obvious benefit is that the drug delivery device can respond to changes in the biomarker in real-time. In addition, it gives much better insight than measuring only the quantity and time of dose taken because the patient and healthcare professional can see how the body responds to the drug. Perhaps the diagnostics could be used to identify more subtle problems. For example, if outcomes were poor in a certain area, an investigation might reveal poor training by the local healthcare facility.
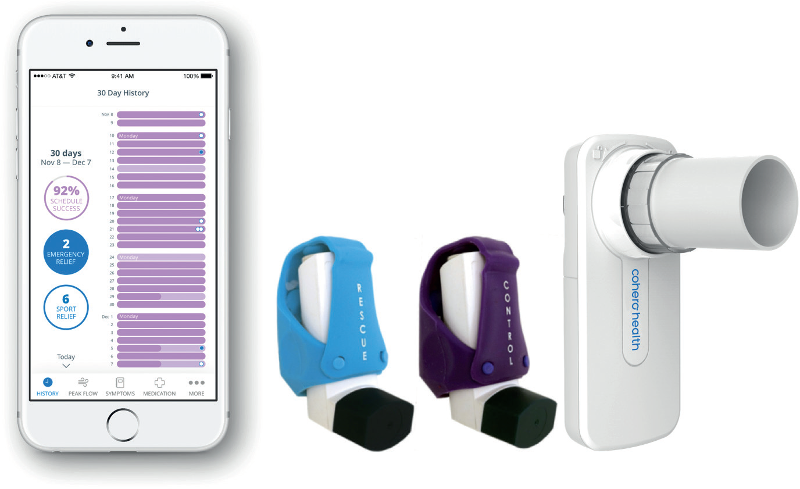
Cohero Health’s BreatheSmart™ app, HeroTracker™ sensors, and spirometer.
Inspair measures inspiratory flowrate so can determine both if the patient’s breathing profile is correct, and if they actuated the inhaler at the right point in their inspiration. Devices which provide feedback like this can be used for ‘continuous improvement’ training, which is more interactive and personalised than a static training video or patient instruction leaflet. It helps with the ‘learn-ability’ of the device, which was identified as a major challenge in recent industry-wide research performed by Springboard.

Biocorp Inspair
However, it is important to consider that add-ons have the obvious disadvantage that the existing geometry is not modified so:
- Add-ons cannot be integrated into the existing device, and thus add bulk to the device, and there are additional use steps to attach them.
- Technical possibilities are limited. For example, it is more difficult to sample the air flow.
Upgrades
A strategy to overcome the limitations of the add-ons is to upgrade an existing design. Novartis is supporting the Propeller add-on [3], but is also upgrading the Breezhaler to the cloudhaler and adding built-in connectivity by working with Qualcomm Life. [4, 5]

Novartis Cloudhaler
H&T Presspart is working with Cohero to upgrade standard Metered Dose Inhaler actuators with connectivity. [6] A notable feature of the Presspart strategy is that the connected functionality is optional. That is, the drug delivery and dose counter functions are still entirely mechanical, so the patient can, in principle, use the device safely and effectively even if the electronic functions fail.

H&T Presspart eMDI
An alert reader will notice that the upgraded devices do not change the use steps or core functionality of the devices. If we want to use electronics and connectivity to change fundamentally the way the patients (and carers, trainers etc.) interact with their device, we need to build the electronics and connectivity into the device from the ground up.
Built-in connectivity
Devices which have connectivity built into them from the beginning can make significant changes in user interaction. The design being built-in also allows more substantial changes to the device, such as advanced features and sensing options. For example, the 3M Intelligent Control Inhaler actively adapts the flowrate to suit the patient and delivers the dose automatically at the right point in the inspiration. A similar example is Opko’s Inspiromatic active dry powder inhaler, and Aptar is partnering with Propeller to develop a new connected metered dose inhaler. [7]
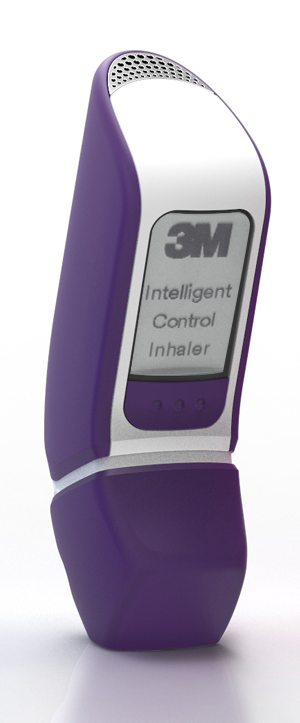
3M Intelligent Control Inhaler
The disadvantages of built-in connectivity mirror substantially the advantages of add-ons. For example, a device with built-in connectivity could be more difficult to make reusable, which adds built-in cost and environmental impact.
Uptake and retention of connected devices
Public interest in connected drug delivery devices has not been measured on a wide scale yet, but we can draw inferences from health-related smartphone apps. More than 50% of US smartphone users downloaded a health-related app in 2015. Individuals more likely to use health-related apps tended to:
- Be younger,
- Have higher incomes,
- Be more educated,
- Be Latino/Hispanic, and
- Have a body mass index (BMI) in the obese range (all P<0.05). [8]
However, weekly retention of apps is poor, even for apps which tend to be associated with hardware such as Fitbit, Garmin and Nike+.
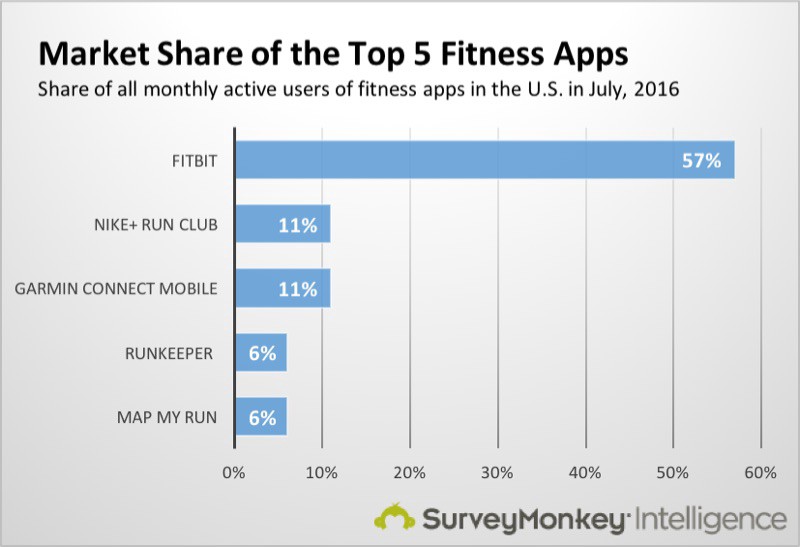
Market Share of the Top 5 Fitness Apps
The reasons for people stopping the use of health apps were primarily:
- High data entry burden 45%
- Loss of interest 41%
- Hidden costs 36%
Out of 1604 people, 662 (41%) said they would not pay anything for a health app. [8]
In November 2017, an analyst from Ernst & Young identified the following hurdles to the widespread adoption of connectivity in drug delivery devices: [9]
- Devices and services.
- Evidence.
- Data infrastructure.
- Business model.
Let us assess the progress and challenges in each area in turn.
Devices and services
Merck Group launched easypod for human growth hormone in 2006, which gained connectivity in 2012, [10] and RebiSmart for multiple sclerosis, which was launched in 2007 and gained connectivity in 2011. They use the easypod Connect [11] and MSdialog [12] web platforms, respectively, for uploading patient data from the device and adding supporting information manually.
One of the challenges is gaining regulatory approval of medical devices that connect to smartphones, but AliveCor has pioneered the way here by being the first to gain FDA approval for smartphone-based medical device software with its atrial fibrillation diagnostic app.
In diabetes, Roche has bought the mySugr web platform, whose Bolus Calculator has Class IIb approval in the EU, and the logbook has Class I approval in both the EU and USA.
So, we can see that connected drug delivery devices are breaking through onto the market, and the regulatory approval and services around them are entirely feasible.
Evidence
Payers and regulators (not to mention patients and healthcare professionals) will want to see clinical evidence for the efficacy of connectivity. Fortunately, the evidence is mounting. Data from Propeller Health, for example, shows reduced short-acting beta agonist use, [13] reduced hospitalisations and reduced emergency room visits. [14] Adherium claims similar clinical evidence. [15]
If efficacy is proven for certain indications, we would still need evidence for preference and adherence. Fortunately, several studies have been done in these areas too. For example:
- An observational study on the RebiSmart device found that 91% liked using the device, and 96% found it ‘easy or very easy to use’. [16]
- An autoinjector preference patient survey found that 82% of BetaConnect patients were ‘highly satisfied’ compared to 67% of RebiSmart and 60% of ExtaviPro patients. [17] The first two devices have connectivity, but the latter does not.
- A retrospective adherence study on patients with multiple sclerosis using RebiSmart found ‘greater than 95% adherence’ over a 140-week duration (N = 110). [18]
Unfortunately, evidence of malware and hacking has also appeared. For example, remote hacking exploits have been demonstrated on some Medtronic insulin pumps, [19] Hospira infusion pumps, [20] and Animas OneTouch Ping insulin pumps. [21]
Data infrastructure
It is logical for companies to roll out their connectivity infrastructure using the following building blocks:
- Adding connectivity to the drug delivery device.
- Optionally connecting the drug delivery device to a ‘mobile medical app’. This can be an app running on a smart phone (such as AliveCor’s KardiaMobile app), or on a dedicated device (such as Abbot’s FreeStyle Libre device).
- Cloud storage and web apps that can be accessed through a web browser.
- Electronic Health Records.
The main cloud computing providers (Amazon Web Services, Google Cloud Platform and Microsoft Azure) have various offering that are HIPAA compliant, so can be used for some medical data in the United States, but HIPAA compliance is not regarded as strong enough protection for the data of EU citizens. The General Data Protection Regulation (in force from 25 May 2018) places further requirements on the controllers and processors of personal data.
Several companies and collaborations are creating cloud technologies to handle data from connected medical devices. For example, Salesforce.com (in the form of its force.com platform), Qualcomm Life and Philips HealthSuite are working on patient data platforms. Roche bought Flatiron which developed the OncologyCloud, claimed by them to be the ‘industry-leading electronic medical record for oncology, advanced analytics, patient portal and integrated billing management’, [22] Medicom is handling the data services for Bayer’s BetaConnect device, and Redox is developing a way to share healthcare data between heterogenous technologies. In effect, Redox can take data from any input, perform transformations and analytics on it, and convert it into a given Electronic Health Record format.
Business models
Payers are trying to reduce costs, so simply adding connectivity and expecting to be able to charge more is not a convincing strategy. However, a holistic view of the health economics of a given indication can reveal compelling business models in some cases.
Example business models already in use include:
- A collaboration between Amgen and Humana whereby Amgen analyses real-world evidence from Humana’s members with data from wearable devices, apps and smart drug delivery devices. [9]
- A collaboration between Amgen and Harvard Pilgrim Healthcare whereby Amgen will fully refund the cost of Repatha if the patient is hospitalised by a stroke or myocardial infarction. Naturally, Harvard Pilgrim will need to show that the patient had adhered to the Repatha regimen and connectivity is a convincing way to do this.
- Abbot did not get reimbursement initially when it developed the FreeStyle Libre flash glucose sensor, so sold it direct to patients. It has since been approved for purchasing by the UK National Health Service. [23]
Summary and final thought
From what Springboard sees in its day-to-day work, every company involved in drug delivery devices either has a connected technology or is developing one. Clinical evidence for improved adherence exists, and evidence for other clinical benefits is mounting. The data infrastructure exists, although systems are not familiar to patients or healthcare professionals yet, and there are legal hurdles to overcome when transferring patient data between legal jurisdictions. Traditional business models have struggled, but innovative business models are progressing.
Connected drug delivery devices are already with us, and more are coming to market. The idea that connectivity itself will solve the adherence problem is unrealistic. However, for the first time, healthcare professionals will be able to identify who is adherent and who is not, which will allow them to refocus efforts on those who have the most difficulty adhering.
If you are from a pharmaceutical company or medical device manufacturer and wish to talk with us at Springboard about any medical device development questions, please get in touch.
References
[1] Iuga AO, McGuire MJ, “Adherence and health care costs”. Risk Manag Healthc Policy, 2014, Vol 7, pp 35–44.
[2] Forissier T, Firlik K, “Estimated Annual Pharmaceutical Revenue Loss Due to Medication Nonadherence”. Capgemini and HealthPrize white paper, 2016.
[3] Tyer D, “Novartis Signs ‘Connected Inhaler’ Deal with Propeller Health”. PMLive, Feb 2017. Accessed May 2018
[4] “Network Connected Inhaler for COPD”. myAirCoach. (Accessed May 2018)
[5] Pai A, “Novartis, Qualcomm Life to Develop Connected Inahler for COPD”. MobiHealthNews, Jan 2016. (Accessed May 2018)
[6] H&T Presspart eMDI product webpage. (Accessed May 2018)
[7] Comstock J, “Propeller Health, Aptar Partner to Create Connected Metered Dose Inhalers”. MobiHealthNews, Feb 2016. (Accessed May 2018)
[8] Krebs P, Duncan DT, “Health App Use Among US Mobile Phone Owners: A National Survey”. JMIR mHealth uHealth, Nov 2015, Vol 3(4), epub.
[9] Handschuh T, “Smart Devices – How to Unlock Their Potential in the Real World.” Presentation at PDA Universe of Prefilled Syringes and Injection Devices, Vienna, Nov 2017.
[10] “Merck Serono Launches easypod™ Connect in Europe”. Merck Serono press release, Oct 2011. (Accessed May 2018)
[11] easypod® Connect product webpage. (Accessed May 2018)
[12] RebiSmart MSdialog product webpage. (Accessed May 2018)
[13] Merchant RK, Inamdar R, Quade RC, “Effectiveness of population health management using the Propeller Health Asthma Platform: A randomized clinical trial”. J Allergy Clin Immunol Pract, May-Jun 2016, Vol 4(3), pp 455–463.
[14] Merchant RK et al, “Interim results of the impact of a digital health intervention on asthma healthcare utilization”. J Allergy Clin Immunol, Feb 2017, Vol 139(2), p AB250.
[15] “The Medication Nonadherence Epidemic”. (Accessed May 2018)
[16] D’Arcy C et al, “Patient assessment of an electronic device for subcutaneous self-injection of interferon ß-1a for multiple sclerosis: an observational study in the UK and Ireland”. Patient Prefer Adherence, Jan 2012, Vol 6, pp 55–61.
[17] Limmroth V et al, “Autoinjector preference among patients with multiple sclerosis: results from a national survey”. Patient Prefer Adherence, Aug 2017, Vol 11, pp 1325–1334.
[18] Solsona E et al, “Impact of adherence on subcutaneous interferon beta-1a effectiveness administered by RebiSmart® in patients with multiple sclerosis”. Patient Prefer Adherence, Mar 2017, Vol 11, pp 415–421.
[19] Goodin D, “Insulin pump hack delivers fatal dosage over the air“. The Register. Accessed July 2018.
[20] Finkle J, “FDA warns of security flaw in Hospira infusion pumps.” Reuters. Accessed July 2018.
[21] Finkle J, “J&J warns diabetic patients: Insulin pump vulnerable to hacking.” Reuters. Accessed July 2018.
[22] Kewon A, “Drug Giant Roche Buys Former Google Execs’ Flatiron in $1.9B Deal”. BioSpace, Feb 2018. (Accessed May 2018)
[23] Woodfield J, “FreeStyle Libre to be available on the NHS from November”. Diabetes.co.uk, Sep 2017. (Accessed May 2018)